The EU AI Act (Regulation (EU) 2024/1689) applies to AI-powered medical devices automatically classified as high-risk under Annex III. Manufacturers of SaMD with AI components must comply by August 2026, or August 2027 for CE-marked devices subject to Notified Body review under MDR or IVDR. Both frameworks apply simultaneously, requiring integrated technical documentation, data governance, and human oversight mechanisms.
This is not a distant compliance horizon. The provisions most relevant to medical device AI became applicable from August 2026. Companies that have not yet assessed their AI systems against the AI Act risk gaps in their technical documentation and conformity processes at exactly the moment Notified Bodies are beginning to incorporate AI Act considerations into their assessments.
This guide explains what the AI Act requires from SaMD developers, how it interacts with MDR and IVDR, and what practical steps manufacturers should be taking now.
For general SaMD MDR compliance, see our SaMD EU MDR Compliance Guide
Timeline Update: April 2026
Since this article was originally published, two important milestones have passed and one critical deadline distinction has been clarified. If you are planning your AI Act compliance roadmap as a SaMD developer, your timeline may need adjusting.
February 2026: Commission published guidelines on Article 6
The European Commission met its deadline under Article 6(5) of the AI Act and published practical guidelines on how to classify AI systems as high-risk, including concrete examples of what qualifies and what does not. If your team has not yet reviewed these guidelines as part of your classification assessment, this is a priority action, they directly affect how you document your high-risk determination in your technical file.
2 August 2026: Most high-risk AI obligations now apply, but with a critical exception for medical devices
As of 2 August 2026, the majority of high-risk AI obligations under the AI Act entered into application. However, there is an important split that is directly relevant to SaMD developers:
Article 6(1) the provision that covers AI systems embedded in products subject to third-party conformity assessment under EU harmonisation legislation, does not apply until 2 August 2027. This is the provision that governs CE-marked medical devices and IVDs regulated under MDR and IVDR, where Notified Body involvement is required.
In practical terms, this means that manufacturers of Class IIb/III medical devices (MDR) or Class C/D IVDs (IVDR) that incorporate AI have until August 2027, not August 2026, to comply with the Article 6(1) high-risk classification pathway. The 2026 deadline applies primarily to AI systems listed in Annex III of the AI Act that are not subject to Notified Body conformity assessment under other EU legislation.
There is also a transitional provision worth noting: AI systems already placed on the market before 2 August 2026 are only subject to the new obligations if they undergo significant design changes after that date.
The full official timeline is available at the EU AI Act implementation timeline.
Revised compliance milestones for SaMD developers
- February 2025: AI literacy requirements and prohibitions on unacceptable-risk AI applied
- August 2025: GPAI model obligations applied (relevant if your SaMD is built on a foundation model)
- February 2026: Commission guidelines on Article 6 classification published
- 2 August 2026: Most high-risk AI obligations apply (Annex III systems without NB assessment)
- 2 August 2027: Article 6(1) applies: key deadline for CE-marked MDR/IVDR SaMD
We recommend beginning compliance preparation now regardless of which deadline applies to your device. Notified Bodies are already incorporating AI Act expectations into their MDR and IVDR assessments ahead of the legal deadline, and the documentation work involved, training data governance, bias assessment, human oversight design, takes considerable time to complete properly.
The rest of this article describes what the high-risk AI obligations require in practice. Read it with the above timeline in mind.
1. Does the AI Act Apply to Your Software?
The AI Act applies to AI systems placed on the market or put into service in the EU. An AI system is defined as a machine-based system that, given explicit or implicit objectives, infers from inputs how to generate outputs such as predictions, content, recommendations, or decisions that can influence real or virtual environments.
This definition is intentionally broad. It covers:
- Machine learning models (supervised, unsupervised, reinforcement learning)
- Deep learning systems including convolutional neural networks used in medical imaging
- Natural language processing tools used in clinical documentation or decision support
- Bayesian classifiers and other probabilistic inference systems
It does not cover:
- Traditional rule-based software with no learning or inference component
- Software that executes fixed logic without adaptive behaviour
If your SaMD uses any form of machine learning or statistical inference to generate clinical outputs, the AI Act almost certainly applies.
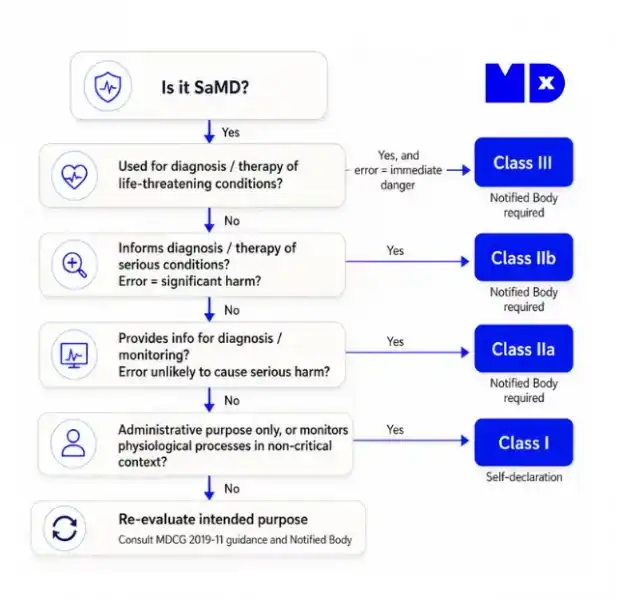
2. High-Risk AI Classification for Medical Devices
The AI Act categorises AI systems by risk level. For medical device manufacturers, the critical category is high-risk AI.
Under Annex III of the AI Act, AI systems intended to be used as safety components of medical devices, or which are themselves medical devices regulated under MDR or IVDR, are automatically classified as high-risk AI.
This means: if your SaMD is a CE-marked medical device or IVD, or is a software component that performs a safety function within one, it is high-risk AI under the AI Act. There is no further classification analysis required, the medical device status determines it.
High-risk AI systems are subject to the full obligations of the AI Act, including:
- Risk management system: an AI-specific risk management process, documented and integrated with the ISO 14971 risk management already required under MDR
- Data and data governance: training, validation, and testing datasets must be relevant, representative, free of errors, and sufficiently complete; demographic and geographic representativeness must be documented
- Technical documentation: a detailed record of the AI system’s design, development process, training methodology, validation approach, and performance characteristics
- Transparency and instructions for use: users must be provided with clear information about the AI system’s capabilities, limitations, accuracy metrics, and circumstances under which human oversight is required
- Human oversight: the system must be designed to allow human oversight and intervention; it must not undermine the ability of the operator or user to override, disregard, or reverse outputs
- Accuracy, robustness, and cybersecurity: performance must be declared and validated; the system must be resilient to errors, faults, and adversarial manipulation
- Conformity assessment: high-risk AI systems must undergo a conformity assessment before being placed on the market
3. How the AI Act Interacts with MDR and IVDR
This is where the compliance picture becomes complex, and where early planning pays off.
The AI Act does not replace MDR or IVDR. Both regulatory frameworks apply simultaneously to AI-powered SaMD. However, the EU has designed a streamlined pathway for medical devices that are already subject to Notified Body review under MDR or IVDR.
Under Article 11 and Annex II of the AI Act, AI systems that are regulated as medical devices benefit from a single technical documentation approach meaning the AI Act technical documentation requirements can be integrated into the existing MDR/IVDR technical file rather than creating a separate document set.
Similarly, for Class IIb and III medical devices (MDR) and Class C and D IVDs (IVDR) which are the most likely to contain high-risk AI the Notified Body involvement already required under MDR/IVDR can cover the AI Act conformity assessment. The Notified Body acts as the relevant conformity assessment body for both frameworks.
In practice this means:
What changes for AI-powered SaMD under the AI Act:
- Technical documentation must now explicitly address AI-specific elements: training data governance, model validation across subgroups, bias assessment, explainability approach, and human oversight mechanisms
- Post-market monitoring must include AI performance monitoring tracking model drift, accuracy degradation over time, and distribution shift in real-world data
- Transparency obligations require new IFU content describing AI limitations and human oversight requirements
- A fundamental rights impact assessment may be required for certain high-risk AI applications in healthcare
What does not change:
- The MDR/IVDR conformity assessment route remains the primary pathway
- The Notified Body relationship established for MDR/IVDR CE marking remains the relevant body
- ISO 14971 risk management, IEC 62304 lifecycle management, and clinical evaluation requirements are unchanged AI Act risk management is additive, not a replacement
4. General Purpose AI (GPAI) Models in Medical Devices
A separate and increasingly relevant category is General Purpose AI (GPAI) large foundation models or multimodal AI systems that can be adapted or fine-tuned for specific applications.
If a SaMD developer is building on top of a GPAI model: for example, fine-tuning a large language model for clinical documentation, or adapting a vision foundation model for medical image analysis both the GPAI model provider and the SaMD developer have obligations under the AI Act.
GPAI model providers must publish technical documentation and comply with copyright and transparency requirements. SaMD developers who deploy or fine-tune GPAI models are responsible for ensuring the resulting system meets all high-risk AI obligations, including data governance, validation, and clinical performance claims. The validation methodology for fine-tuned GPAI models in medical contexts is an area where regulatory guidance is still developing, early engagement with your Notified Body is strongly recommended.
5. Key Timelines
August 2024: AI Act enters into force.
February 2025: Prohibitions on unacceptable-risk AI systems apply. Not directly relevant for medical SaMD, but important for any AI used in patient-facing administrative processes.
August 2025: GPAI model obligations apply. SaMD developers building on foundation models must assess their exposure now.
August 2026: High-risk AI obligations fully apply. This is the key deadline for medical device AI. From this date, new AI-powered SaMD placed on the EU market must comply with all high-risk AI requirements.
Post-2026: Notified Bodies designated under the AI Act will begin conducting AI Act-specific conformity assessments. The intersection with MDR/IVDR NB assessments will become a standard part of the conformity process.
6. What to Do Now: A Practical Checklist
Classify your AI systems. Identify every AI component in your SaMD portfolio and confirm whether it meets the EU’s definition of an AI system. For each, document the risk classification and the rationale.
Assess your technical documentation gaps. Review your existing MDR/IVDR technical files against the AI Act Annex IV requirements. Identify where AI-specific content, training data documentation, bias assessment, explainability approach, is missing or insufficient.
Review your data governance. The AI Act’s requirements for training data representativeness and bias documentation are more explicit than anything in MDR. If your training data governance is not documented at the level the AI Act requires, this is a gap that needs addressing before your next Notified Body audit.
Update your IFU and labelling. Transparency obligations mean users must be explicitly informed about AI limitations, performance metrics across relevant subgroups, and circumstances requiring human override. Most current SaMD IFUs are not written to this standard.
Engage your Notified Body. Ask your NB directly how they are approaching AI Act integration into MDR/IVDR assessments. Different NBs are at different stages of readiness, and early clarity on what they will expect prevents last-minute documentation gaps.
Build AI performance monitoring into your PMS. Post-market surveillance for AI-powered SaMD must now track model performance over time. If your PMS plan does not include AI-specific monitoring metrics, update it before August 2026.
Read more about Software, Digital Health and AI services.
Key Requirements at a Glance
AI-powered SaMD classified as high-risk under the EU AI Act must meet the following obligations before market placement:
- Risk management system: An AI-specific risk management process, documented separately from but integrated with ISO 14971 requirements under MDR/IVDR.
- Data governance: Training, validation, and test datasets must be representative, bias-assessed, and documented for demographic and geographic coverage.
- Technical documentation: AI-specific content, including training methodology, model validation, and performance characteristics, must be incorporated into the MDR/IVDR technical file.
- Human oversight: The system must be designed so clinicians can override, disregard, or reverse AI outputs at all times.
- Transparency and IFU: Instructions for use must explicitly state AI limitations, accuracy metrics, and when human review is required.
- Conformity assessment: CE-marked SaMD undergoes AI Act conformity assessment through the existing Notified Body route (Article 6(1), applicable from August 2027).
AI Act SaMD Classification: When Is Your Software Automatically High-Risk?
Under the EU AI Act, Software as a Medical Device does not go through a separate AI risk classification process if it is already regulated as a medical device. The classification is automatic.
The rule under Annex III: Any AI system that is itself a medical device, or functions as a safety component of a medical device, regulated under EU MDR or IVDR, is automatically classified as high-risk AI under Article 6(1).
- A Class IIa diagnostic imaging AI — high-risk AI.
- A Class III surgical decision-support algorithm — high-risk AI.
- A Class C IVD with an AI-based interpretation engine — high-risk AI.
- Rule-based software with no learning component — not covered by the AI Act.
EU AI Act August 2026 Deadline: What It Means for Medical Device Manufacturers
The date most frequently cited, 2 August 2026 is real, but it does not apply equally to all medical device AI. Understanding the split is critical for compliance planning.
What applies from 2 August 2026
High-risk AI obligations apply to Annex III AI systems not subject to third-party conformity assessment. In the medical device context: hospital workflow tools, patient triage algorithms not CE-marked as medical devices.
What applies from 2 August 2027
For CE-marked MDR/IVDR devices, Article 6(1) applies and it does not enter into force until 2 August 2027. This is the deadline that matters most for SaMD developers.
AI Act vs MDR: Understanding the Dual Compliance Framework for SaMD
AI-powered medical devices are subject to two overlapping EU regulatory frameworks simultaneously. They are not alternatives — both apply, and compliance with one does not fulfil the other.
| EU MDR / IVDR | EU AI Act | |
|---|---|---|
| What it governs | Safety, performance, and clinical efficacy of the device | How the AI was built, trained, validated, and governed |
| Key deadline | Ongoing (MDR transitional periods ended) | Aug 2026 (non-NB AI) / Aug 2027 (CE-marked SaMD) |
General Purpose AI (GPAI) Models in Medical Devices: What the AI Act Requires
A General Purpose AI (GPAI) model is an AI model trained on large amounts of data, capable of serving a wide range of downstream tasks. Foundation models and LLMs adapted for clinical use fall into this category.
Tier 1: GPAI model provider obligations (August 2025)
The company that trains the foundation model must document training data, comply with copyright law, and publish transparency summaries — applicable since August 2025.
Tier 2: SaMD developer obligations (August 2026/2027)
When a SaMD developer fine-tunes a GPAI model for a medical application, they become the high-risk AI provider and must conduct their own clinical validation, document training data for fine-tuning, and design human oversight mechanisms specific to the clinical context.
What is the EU AI Act?
The EU AI Act (Regulation (EU) 2024/1689) is the European Union’s comprehensive legal framework for artificial intelligence systems. It entered into force on 1 August 2024 and is being phased in progressively through 2027.
What does it regulate?
The AI Act classifies AI systems by risk level, from unacceptable-risk (prohibited) to high-risk, limited-risk, and minimal-risk, and sets obligations proportionate to that classification. High-risk AI systems, which include all AI used in medical devices regulated under MDR or IVDR, face the most stringent requirements.
Who does it apply to?
Any organisation that develops, deploys, or places on the EU market an AI system, including manufacturers of AI-powered Software as a Medical Device (SaMD), regardless of where they are based.
How does it differ from MDR?
The MDR governs whether a device is safe and performs as claimed. The AI Act governs how the AI system within that device was built, trained, and governed. Both apply simultaneously to AI-powered SaMD from 2026–2027 onwards.
Frequently Asked Questions: EU AI Act and Medical Devices
The MDR (Medical Device Regulation) governs the safety, efficacy, and quality of medical devices, including those powered by AI. The EU AI Act is a separate regulatory framework that addresses the risks and accountability of AI systems themselves. The AI Act focuses on how the AI system was built, trained, validated, and deployed, while MDR focuses on the clinical performance of the device. Both apply simultaneously to AI-powered SaMD from August 2026 onwards.
Yes, if you use any form of machine learning, deep learning, or statistical inference to generate clinical outputs, the AI Act applies. This includes supervised learning, convolutional neural networks for medical imaging, natural language processing for clinical documentation, and Bayesian classifiers. It does NOT apply to traditional rule-based software with fixed logic and no learning or inference capability
High-risk AI includes AI systems that are themselves medical devices or safety components of medical devices regulated under MDR, or AI systems regulated under IVDR. If your SaMD is CE-marked or classified as a medical device, it is automatically classified as high-risk AI. High-risk AI must comply with all AI Act obligations: risk management, data governance, technical documentation, transparency, human oversight, and conformity assessment.
The AI Act requires explicit documentation that training, validation, and testing datasets are relevant, representative, free of errors and bias, and sufficiently complete. Demographic and geographic representativeness must be documented, particularly important for medical AI to ensure performance across age, sex, ethnicity, and geography. This is more explicit than MDR alone.
High-risk AI systems must be designed to enable human oversight and intervention. Users must be able to override, disregard, or reverse the AI’s decision, and the system must not undermine this ability. For clinical SaMD, this typically means the AI operates in decision-support mode and clinicians retain authority to override recommendations.
August 2026 is the key deadline for medical device AI. From this date, all new AI-powered SaMD placed on the EU market must comply with high-risk AI requirements, Notified Bodies will incorporate AI Act assessments into MDR/IVDR reviews, and technical documentation must include AI-specific content.
Master Artificial Intelligence (AI) in Medical Devices

Join regulatory experts Alberto Bardají (Quixano) and Diego Rodríguez Muñoz (MDx) for a practical live webinar on building evidence for AI-powered medical device compliance.
What you’ll learn:
- Regulatory pathways: How to navigate both MDR and AI Act requirements simultaneously
- Data generation strategies: Building pre-clinical and clinical evidence for AI/ML models
- Bias assessment & documentation: Demographic and geographic representativeness requirements
- Real-world case studies: Lessons from companies navigating the August 2026 deadline
- Best practices: How to integrate AI Act requirements into your existing MDR technical file
Session format: Live interactive Q&A | Duration: 60 minutes | When: Upcoming date via LinkedIn